| Solar-generated fuels show promise as a renewable energy carriers for the future. In the Bernhard lab, our research in this area focuses on searching for new catalysts for photolytic water splitting, light-driven reduction of metal ions, and photo-induced dehydrogenation reactions of bio-molecules. By creating novel inorganic compounds and organo-metallic ligand architectures, we are able to tune the electro-chemistry of our complexes and build supramolecular structures. This approach allows us to arrange mole-cular components energetically and spatially in order to optimize the fuel producing reactions. Reduction cata-lysts are studied individually, with sacrificial species replacing the opposing oxidation reaction, in order to scientifically determine the structure-property relationships significant to the creation of a full solar fuel system. New catalytic systems are evaluated in our home-built photoreactors, which enable us to monitor the kinetics of fuel evolution for multiple reactions simul-taneously and in real time. |
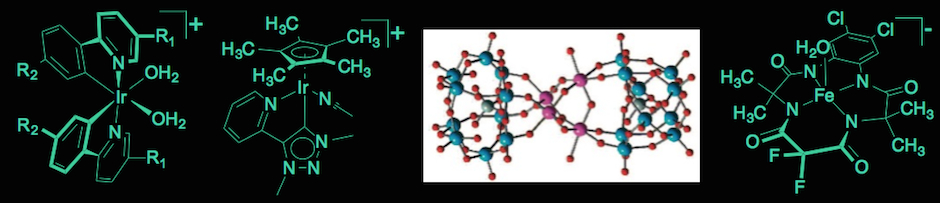
Photocatalytic hydrogen production of a family of Ir(III) photosensitizers with in situ generated colloidal Pt. |
|
In order to develop a complete solar fuel system, it is necessary to replace the sacrificial reductant used in photocatalytic reductions with an efficient water oxidation catalysis process. Our recent efforts have very successfully investigated cyclometalated Ir(III) aquo complexes as robust water oxidation catalysts. By substitution of the cyclometalating 2-phenylpyridine ligands of these complexes, it has been shown that a broad range of oxidation potentials can be achieved. A new collaboration with Martin Albrecht (University College, Dublin) targets families of Ir-based water oxidation catalysts that use strongly bound carbene ligands. An initial publication in Angewandte Chemie highlighted the robustness of these catalysts with the documentation of turn-over numbers approaching 10,000. Currently, follow-up work using similar catalysts is being performed to analyze the catalytic mechanism of these aquo complexes carefully. UV-Vis spectroscopy is employed in combination with oxygen evolution studies to pinpoint reactive intermediates. Dynamic light scattering ruled the presence of colloidal materials out and documented that gas bubbles can easily be mistaken for nano-particulate matter in the reaction solutions. Immobilization of the catalysts on photoelectrodes was used as a stepping stone towards the implementation of a full water splitting system.
Two other catalyst designs were discovered in the Bernhard group. A collaboration with Marcella Bonchio (University of Padova) on polyoxometalates (POMs) yielded a novel, efficient Ruthenium-based catalyst. Recently, work on Fe-TAML complexes and their water oxidation capacity was started in collaboration with the Collins group at Carnegie Mellon. The catalyst exhibits unprecedented turnover frequencies of 1 per second.
Our lab is also involved in the area of organic electronics research that employs both polymers and small molecules as luminophores and charge transport materials. Currently, our main focus is placed on photovoltaic appli-cations. The challenge of this field is achieving self-directed photoinduced charge transfer from an absorbing material through the device layers to the electrodes. The ability to simultaneously control nano-scopic morphology, light absorption, and electronic potential is fundamental to creating a successful solar cell. One of the key aspects of this work is the design of electron transport materials. In the case of transition metal complexes, each of the three key elements are tunable by virtue of the metal’s organic ligand scaffold. Synthetic ligand modification is a specialty in the Bernhard group, and organic photovoltaics is perfectly suited to be its star application. Recent work in collaboration with the Noonan lab focuses on the synthesis of electrochemically and photo-physically active heterocyclic compounds. The main emphasis is placed on electron transport materials. The Bernhard lab evaluates the electron acceptor properties with cyclic voltammetry. Luminescence spectroscopy and spectro-electrochemical techniques are used to further examine the electronic structure of these materials. These properties are modeled by DFT-calculations which are then used for pin-pointing structures with improved properties, closing a structure-activity feedback loop.
|
|
 |
Heterleptic iridium complexes were used for the color tuning of light-light emitting electrochemical cells |
The Bernhard group successfully tuned the color of light-emitting electrochemical cells (LECs), a single-layer variety of OLEDs, that has gathered a lot of recent interest. An attractive extension of our OLED electro-luminophore research is the use of similar chiral metal complexes in 3D display applications. The principle behind stereoscopic displays is to provide the viewer with two different slightly offset images in order to achieve a 3-D effect. Limitations of traditional approaches (e.g. color consistency, expensive filter technology) can be overcome by using circularly polarized light to distinguish between the two stereo images. Circularly polarized luminescence can be observed from chiral molecules, but emission dissymmetry factors are typically small making this approach inapplicable for the design of stereoscopic displays. In order to improve the degree of circularly polarized photoluminescence it is necessary to incorporate chromophores into stable chiral matrices, such as chiral-nematic liquid crystals, preferably with the ability to form stable glasses. Little work has been dedicated so far to using the helicity of coordination complex-based luminophores and extending their chirality to nanometric ensembles. The figure below depicts the progression from a chiral ligand to an electroluminescent device with chiroptical properties. An absorption-based device, e.g. responding to an electrochromic change, can be envisioned correspondingly.
|
|


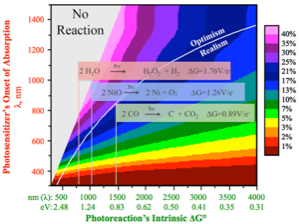 Optimal power efficiency of a solar fuel reaction plotted as a function of both a photoreaction’s intrinsic ΔG° and the photosensitizer’s onset of absorption.
Optimal power efficiency of a solar fuel reaction plotted as a function of both a photoreaction’s intrinsic ΔG° and the photosensitizer’s onset of absorption. Parallel Photoreactors utilize intense LED light for the illumination of solar fuel producing catalytic reactions
Parallel Photoreactors utilize intense LED light for the illumination of solar fuel producing catalytic reactions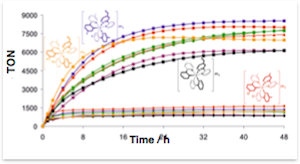
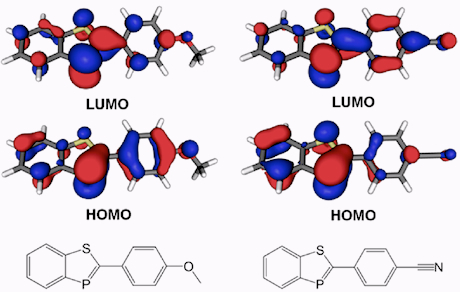 Tuning the electrochemical properties with phosphorus
Tuning the electrochemical properties with phosphorus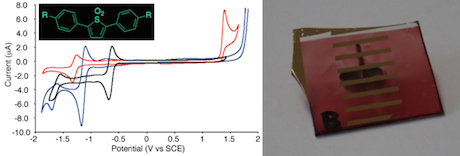 Cyclic voltammetry of an electron transport material (left) Example of a organic photovoltaic cell (right)
Cyclic voltammetry of an electron transport material (left) Example of a organic photovoltaic cell (right)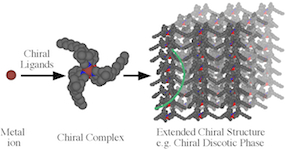 Chiral ligands systems are used to control the helicity of metal complexes. Arrangment of these complexes in large scale ensembles leads to massive emission and absorption dyssymmetries
Chiral ligands systems are used to control the helicity of metal complexes. Arrangment of these complexes in large scale ensembles leads to massive emission and absorption dyssymmetries