|
| Research
• Regioregular Poly(3-alkylthiophene) • End Group functionalization • Side Chain Functionalization • Lab Tour
|
End Group FunctionalizationTypical synthesis of PATs via Rieke, GRIM or McCullough methods produces a polymer with 2 different end groups: H/Br and H/H (PATs made by the McCullough method also contain traces of Br/Br) that cannot be separated from each other. Here, we are interested in controlling the nature of the end groups of HT-PATs. HT-PATs with certain end groups may be very useful for end-group driven self-assembly onto surfaces and into conducting polymer networks, as well as in building blocks for the synthesis of block copolymers.
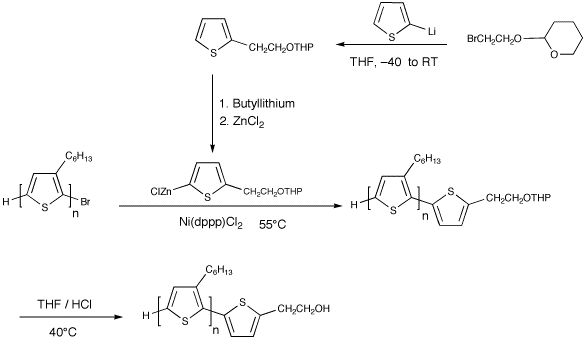
We have investigated two approaches to alter the end group composition of HT-PATs: post-polymerization functionalization and in-situ modification. Post-Polymerization FunctionalizationIn this method, HT-PHT with mainly H/Br end group composition was first made and isolated.1 The bromine end of the polymer was then coupled with a thiophene bearing tetrahydropyranyl protected hydroxyl group. The reaction was monitored by MALDI-TOF MS. The product was reacted with HCl to yield HT-PHT with about 90% hydroxyl groups at one end. An amine group was also similarly incorporated by coupling the bromine end group with a stabase-protected amino group. The amine group incorporation was slightly more difficult to monitor due to loss of the protecting group during the work up. As a result the MALDI contains a mixture of protected amino groups, free amino groups and a small amount of H/H polymer.
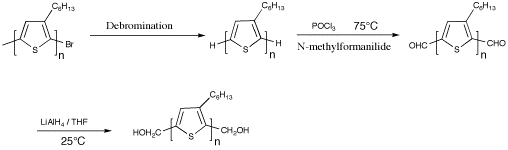
We also incorporated aldehyde groups onto both ends of PAT's via the Vilsmeier reaction of a H/H terminated polymer. PHT was prepared via the McCullough methods to produce a polymer bearing the typical distribution of end groups, H/H and H/Br. The end groups are then all converted to H/H by treating the polymer with excess t-butylMgCl after work up. After debromination, the Vilsmeier reaction is used to introduce aldehyde groups onto the polymer ends. To drive the reaction forward, a large excess of reagents was used at 75 ºC. The Vilsmeier reaction had good selectivity and side reactions on the 4- positions were rarely seen. This reaction was monitored using MALDI-TOF MS. The aldehyde groups could be reduced to create a PAT with hydroxy groups on both ends. The hydroxy terminated polymer was used as building blocks for the synthesis of tri-block copolymers.2 A similar strategy was employed to synthesize amine terminated PAT's for use in hybrid solar cells.3
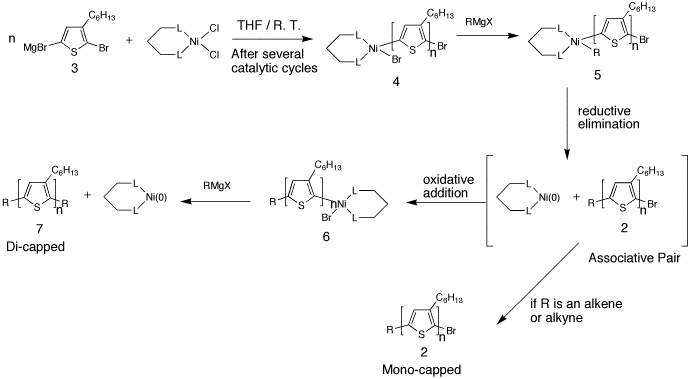
In-Situ ModificationThe first attempt toward in-situ end group modification was reported by Langeveld-Voss et al. A PAT was synthesized using the McCullough methods. The standard work-up was altered by the addition of a second Grignard and some fresh catalyst prior to quenching with MeOH. MALDI-TOF MS analysis of the resultant polymershow that a mixture of different products were obtained (monofunctionalized, difunctionalized and H/H). The major limitation of this report is that it was limited to a trimethylsilyl group and the molecular weights of the polymer were low.4 We have recently developed a more versatile method that does not require to isolate a polymer bearing predominately H/Br end groups. In this approach a PAT is syntheisized using the GRIM method. Prior to quenching the polymerization, a second Grignard reagent is added. This second Grignard reagent effectively terminates the polymerization and “caps” the polymer. This method has been demonstrated to work with a variety of different types of Grignard reagents (i.e., aryl, alkyl, allyl, vinyl, etc.). The reactivity of these depends on their nature, where allyl, ethenyl and vinyl groups produced monofunctionalized polymers and all others yielded difunctionalized polymers.5 The end group composition of the polymers approaches 100% in some cases and can be monitored using a combination of MALDI-TOF and 1H NMR. By utilizing the proper protecting groups -OH, -CHO, and -NH2 groups have been incorporated onto the polymer ends.5 The main advantage of this method is that it allows for the in-situ functionalization of regioregular polythiophene, generating a variety of end-capped polymers in one step.
References:
|
|||||